Can semaglutide dosing characteristics be used to estimate the risk of residual gastric contents?
Imaging is the only way to accurately assess residual gastric contents
With numerous patients taking GLP-1 receptor agonists (GLP-1 RAs), such as semaglutide and tirzepatide, anesthesiologists have sought ways to estimate the risk that these patients have increased residual gastric contents. Unfortunately, outside of imaging, there is no accurate way to quantify that risk.
The American Society of Anesthesiologists (ASA) practice guideline for GLP-1 RAs published in 2024 lists risk factors that may indicate that patients taking these medications have residual gastric contents present.1 The problem with these risk factors is that many patients have residual gastric contents without having any of the listed risk factors present. As we will see, details about the dosing of semaglutide, in particular, offer no insight into the likelihood of having increased residual gastric contents.
To evaluate the incidence of residual gastric contents in patients taking semaglutide, a group of researchers performed a prospective, multicenter, matched study using gastric ultrasound to examine patients prior to surgery. Patients were included if they were taking semaglutide for diabetes or weight management and were undergoing general anesthesia for elective surgery, and had held one dose of the medication. They were matched based on age, diabetes status, and BMI to other patients undergoing elective surgery who were not taking any GLP-1 RAs. Patients with conditions known to delay gastric emptying (e.g., Parkinson disease, hypothyroidism, etc.) were excluded. A total of 88 patients were enrolled (44 in each group) and underwent gastric ultrasound in the preoperative setting.
A full stomach, defined as having ≥ 1.5 mL/kg of gastric volume, was observed in 49% (n = 21) of the patients taking semaglutide and in 18% (n = 8) of the controls (OR 4.29; 95% CI 1.63–11.29, p = 0.003). Solid gastric contents were observed in 85% (18/21) of the semaglutide patients with full stomachs. Notably, these results are in the setting of holding one dose of semaglutide and with a median fasting duration from solids of 14.4 hours in the semaglutide group and 15.1 hours in the controls—far exceeding fasting guidelines.2 This study further supports the notion that holding one dose of GLP-1 RAs does nothing to reduce the risk of residual gastric contents. It also supports the need for longer fasting durations in these patients.
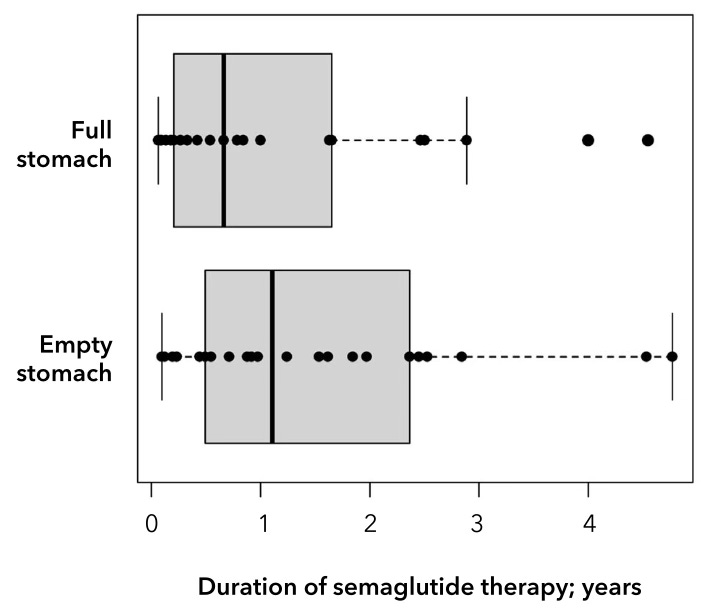
The authors found no significant association between the route of administration (oral vs. subcutaneous), duration of use, dose, or time since the last dose of medication. Even patients who had been taking semaglutide for years still had increased residual gastric contents. I hope this makes clear that the risk factors listed in the ASA practice guideline cannot be used to exclude the risk of having increased residual gastric contents.
One should assume that many, if not most, patients taking GLP-1 RAs have full stomachs. Imaging modalities, with gastric ultrasound being the only feasible option, represent the only way to risk-stratify these patients and exclude the presence of increased residual gastric contents.
For a more in-depth discussion of GLP-1 RAs and their management, see my previous post on the topic.
They include a higher dose of the drug, weekly dosing, being in the escalation phase (as opposed to being on the same dose for several months), and gastrointestinal symptoms such as nausea, to name a few.
The median fasting duration from liquids was > 9 hours in both groups.